|
The bomb was made in the form of a sphere with pieces of plutonium, each below the critical mass, at the edge of the sphere. Recently, Gao 26 calculated the spontaneous ssion half-lives for heavy and super-heavy nuclei by applying a new method of numerically solving Schrdinger equation. Therefore, scientists developed a plutonium-239 bomb because Pu-239 is more fissionable than U-235 and thus requires a smaller critical mass. Normal weapons-grade plutonium contains about 6 percent Pu-240, an isotope with a much higher rate of spontaneous fission (and hence higher neutron generation). the WKB approximation model for spontaneous ssion 20, 21, cold ssion 22, 23 and ternary ssion 24, 25 of super-heavy nuclei and Actinides. When one piece in the form of a bullet is fired into the second piece, the critical mass is exceeded and a chain reaction is produced.Īn important obstacle to the U-235 bomb is the production of a critical mass of fissionable material. The original design required two pieces of U-235 below the critical mass. 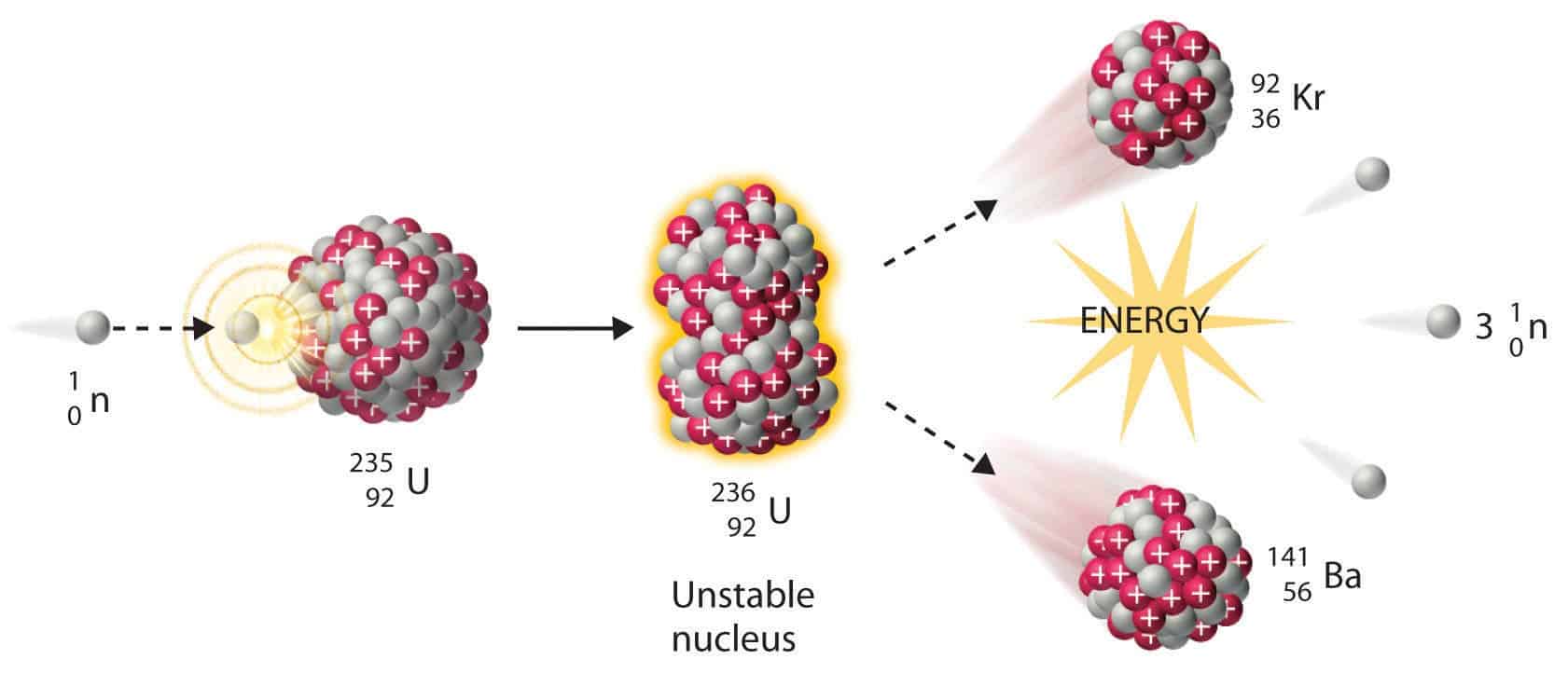
When the critical mass reaches a point at which the chain reaction becomes self-sustaining, this is a condition known as criticality. The minimum mass needed for the chain reaction to occur is called the critical mass. In addition, the uranium sample must be massive enough so a typical neutron is more likely to induce fission than it is to escape. To produce a controlled, sustainable chain reaction, the percentage of U-235 must be increased to about \(50\%\). (These discoveries were taking place in the years just prior to the Second World War and many of the European physicists involved in these discoveries came from countries that were being overrun.) Natural uranium contains \(99.3\%\) U-238 and only \(0.7\%\) U-235, and does not produce a chain reaction. The possibility of a chain reaction in uranium, with its extremely large energy release, led nuclear scientists to conceive of making a bomb-an atomic bomb. Control energy production in a nuclear reactor. View a simulation on nuclear fission to start a chain reaction, or introduce nonradioactive isotopes to prevent one. 
The energy released in this process can be used to produce electricity. \): In a U-235 fission chain reaction, the fission of the m nucleus produces high-energy neutrons that go on to split more nuclei.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
